For its radiopharmaceutical, the sponsor initially conducted labeling experiments but was unable to achieve sufficient radioactivity labeled to their ligand (molar activity) for administration at a therapeutic dose in patients. TRACER optimized the labeling strategy, which increased the molar activity compared to the initial activity. After achieving a level suitable for therapeutic dosing, production started at one of our GMP-certified radiopharmacies to manufacture the study drug suitable for human administration. Once the Phase 1 radiopharmaceutical trial started, the sponsor needed several doses per week to match its patient dosing schedule. By qualifying an additional Iodine-123 supplier, TRACER ensured a reliable supply chain and continuous delivery of the radiopharmaceutical.
Labeling optimization
The labeling yield could be significantly improved by modifying the reaction conditions. The sponsor initially applied standard iodination methods; however, TRACER optimized parameters, such as reaction time, buffer composition, and other process variables. The optimization of the labeling strategy resulted in an increase in achievable molar activity that was suited for clinical doses.
In essence, common radiolabeling methods are not always optimal for every scenario. Specialized experience is essential to identify limiting factors and refine reaction conditions accordingly.
CMC work and GMP implementation
Following optimization of the labeling strategy and method, a GMP-grade drug product was developed in compliance with regulations and clinical trial requirements. The provided CMC activities included:
- Defining and validating all production steps of the radiolabeled compound
- Establishing analytical methods and quality control strategies
- Documenting the full manufacturing process
The CMC documentation was reported and included in the IMPD, which was submitted for regulatory approval in the UK.
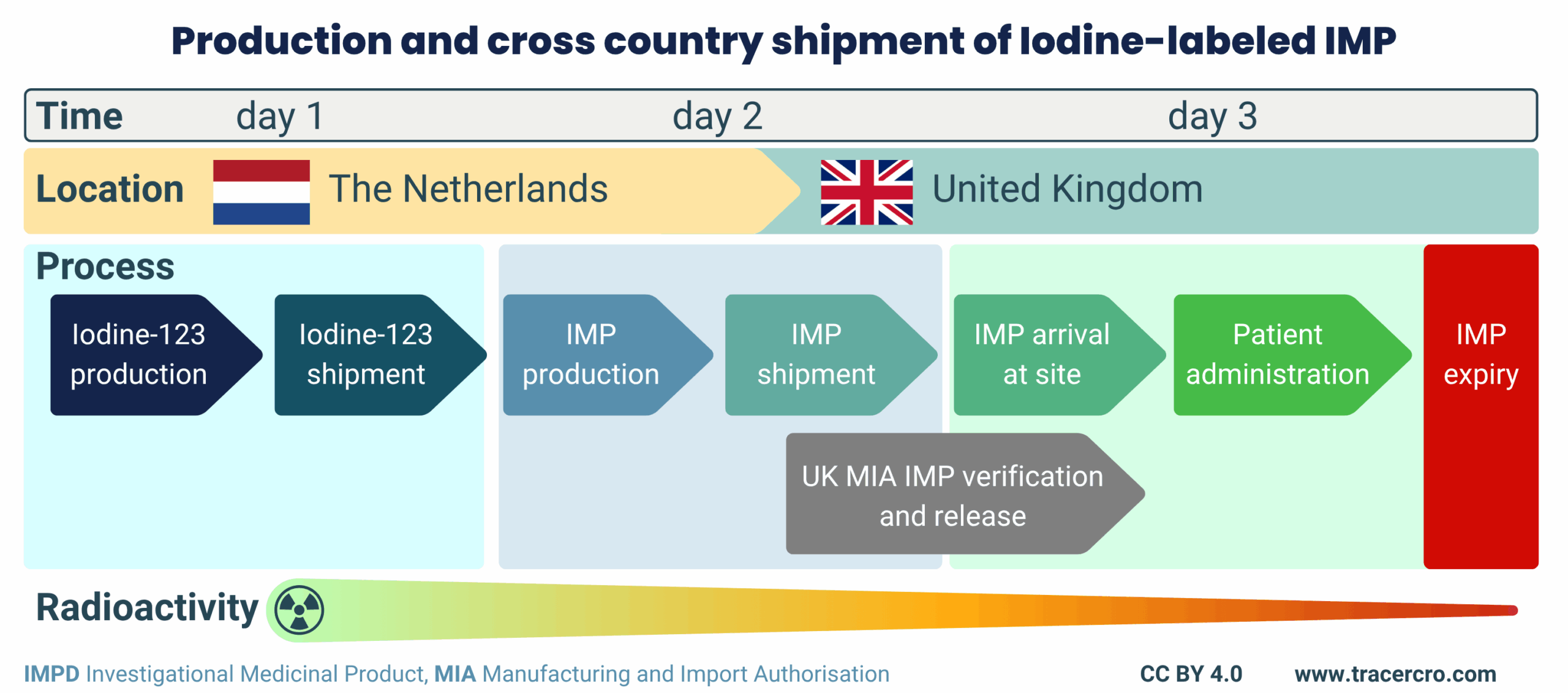
Radiopharmaceutical logistics
The drug product was manufactured in the Netherlands and shipped to the UK for clinical use. TRACER was responsible for both production and logistics. Given the short half-life of Iodine-123 (13.2 hours), scheduling was critical. TRACER successfully delivered the isotope to the radiopharmacy, completed manufacturing and provided QP release. Shipment of the final product was completed within two half-lives. As a result, sufficient activity, in the high MBq to GBq range at the time of administration, was maintained for the highest dose level.
Reliability of delivery regarding multiple administrations
The patients participating in this study received multiple administrations over a period of several weeks. To ensure the evaluability of a patient, all doses needed to be administered according to the schedule. This made the reliability of supply essential. A missed batch could result in a patient becoming non-evaluable, potentially impacting the study timeline and data integrity.