The sponsor developed a novel small molecule for a newly discovered pan-cancer target. TRACER designed a first-in-human microdose SPECT (Single Photon Emission Computed Tomography) imaging study to verify, based on tumor uptake, that the ligand was internalized via its target-based pathway. TRACER also developed the radiolabeling strategy for the investigational ligand to study whole-body distribution and quantify tumor uptake with SPECT.
Primary study research question
“What is the biodistribution, and is there tumor-specific uptake?”
The SPECT imaging study should show the ligand’s tumor targeting ability and any potential off-target binding. By including multiple indications, an assessment of which indication demonstrates the best binding can be made.
Methods
SPECT/CT, planar imaging, blood samples, and, in case of surgery, histopathology analysis for target identification.
Flexible study design to prevent amendments
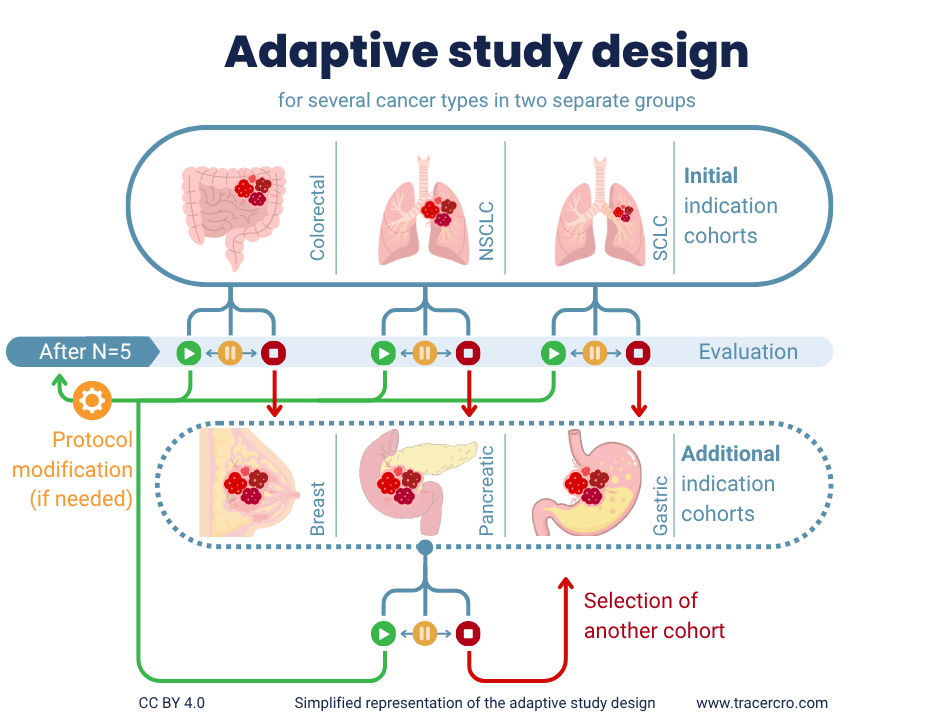
To ensure flexibility during clinical execution, TRACER’s physician scientists and medical liaisons wrote an adaptive study design. In this case, it included three initial cohorts (colorectal, small cell, and non-small cell lung cancer) with room to add other indications. This design reduces the need for substantial modifications or amendments from competent or ethical authorities. Resulting in limiting delays in clinical execution. The study design was approved by the institutional review board (IRB) in about 60 days.
How does this Bayesian study design work?
With a Bayesian study design, incoming data can be used for continuous updates. In this case, an interim analysis of subgroups improves resource utilization. After N=5 per cohort, the cohort will be evaluated by an internal board of specialists based on predefined endpoints and conditions regarding safety, pharmacokinetics (PK), biodistribution, and imaging data.
The decision could be made to:
- continue enrolment with 5 more participants (in case of positive results);
- a cohort could be placed on hold (to discuss arising issues that could lead to stopping enrollment for the indication completely);
- modify protocol regarding the imaging schedule;
- stop further enrolment of an indication based on safety, PK, biodistribution and imaging data (in case of safety concerns, enrollment or assessment issues).
When stopped, an additional cohort (breast-, pancreatic, or gastric carcinoma) could be opened. This study design allows for adding indications without having to do another submission or even a study amendment.
Multi-site study
The study design was for a multicenter trial where the various sites were included in the initial submission. By including multiple centers from the start, you get competitive recruitment. Overall, this means faster accrual rates and a lower chance of delays. To secure standardized results, all (dosimetry) data was analyzed centrally.