What’s the current state of AI for drug discovery? Artificial intelligence (AI) and machine learning (ML) are revolutionizing many fields, and pharma is no exception. AI drug development is accelerating the discovery and development of therapies. From AI drug discovery, to drug repurposing, here we list current and future use cases. AI and drug discovery are a great match: artificial intelligence makes it possible to interpret and recognize patterns in large amounts of data, which would otherwise be virtually impossible. But with the many drug candidates found through virtual screening, we need fast go/no-go decisions to prevent the drug development system, where resources are already scarce, from becoming overloaded.
Fast clinical validation for AI-discovered drugs
TRACER offers a solution based on the Phase 0/early Phase 1 framework that allows fast clinical, in-patient validation of drug candidates. This is particularly useful for AI drug discovery, where the multitude of newly identified targets and compounds requires rapid validation. TRACER is unique in integrating molecular imaging methods into clinical trials. With nuclear imaging methods such as positron emission tomography (PET), whole-body distribution and on and off-target binding can not only be visualized, but also quantified over time. TRACER delivers the data needed to substantiate AI hypotheses regarding new targets, compounds, proteins, and their predicted behavior. You can schedule a meeting to learn more.
Schedule your meeting
AI for drug discovery is changing how drugs are discovered
Can AI be used in drug discovery? AI for drug discovery is not just a tool, it digitizes the field and fundamentally changes the way drugs are discovered. Pattern recognition leads to new targets and biomarkers, and with that, millions of compounds can be screened virtually. The examples below, with the platforms used, are real AI in drug discovery examples.
- Based on routine clinical data, new biomarkers can be found for undiagnosed diseases (Milton AI)
- Gene expression and modulation can be used to discover what’s driving disease and how to bring cells back to a healthy state (Algen Biotechnology, AlphaGenomics, AlphaMissense, General Expression Transformer (GET) from Columbia University)
- Protein structure and interaction can be predicted (AlphaFold, OpenFold, SeedFold)
- From drug discovery to drug formulation and analysis with AI-run robotic labs (Iktos and Insilico)
- Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) can be predicted (ADMET-AI)
Why this article on AI drug discovery and development?
At this moment, compounds discovered with AI are already in clinical trials. AI in drug development is a rapidly growing field with an over 60% year-over-year compound growth [1].
Most major pharmaceutical companies already work with AI partners for target and compound discovery. And vice versa, tech companies are stepping into the pharmaceutical industry. The most remarkable example is ByteDance, the company behind the social media app TikTok, scrolling through the possibilities of AI for drug discovery. But what impact does AI at the moment already have on drug discovery and development? After explaining the basics, this article describes examples and lists the top AI drug discovery companies and the pharmaceutical companies they work with.
Basics: How does AI in drug discovery and development work?
The building blocks of AI consist of data, computational power (for machine learning (ML) and the more advanced deep learning (DL), and algorithms. A great variety of datasets can be used, topics like DNA, disease biomarkers, protein folding, multi-omics, medical imaging, patient routine clinical data, literature, patents, experiments, and many more. These datasets can be used to train AI to improve recognition of known characteristics and relationships, which can then be used for predictability, like protein folding, target binding, drug repurposing, and absorption, distribution, metabolism, excretion, and toxicity (ADMET).
ADMET prediction with ADMET-AI
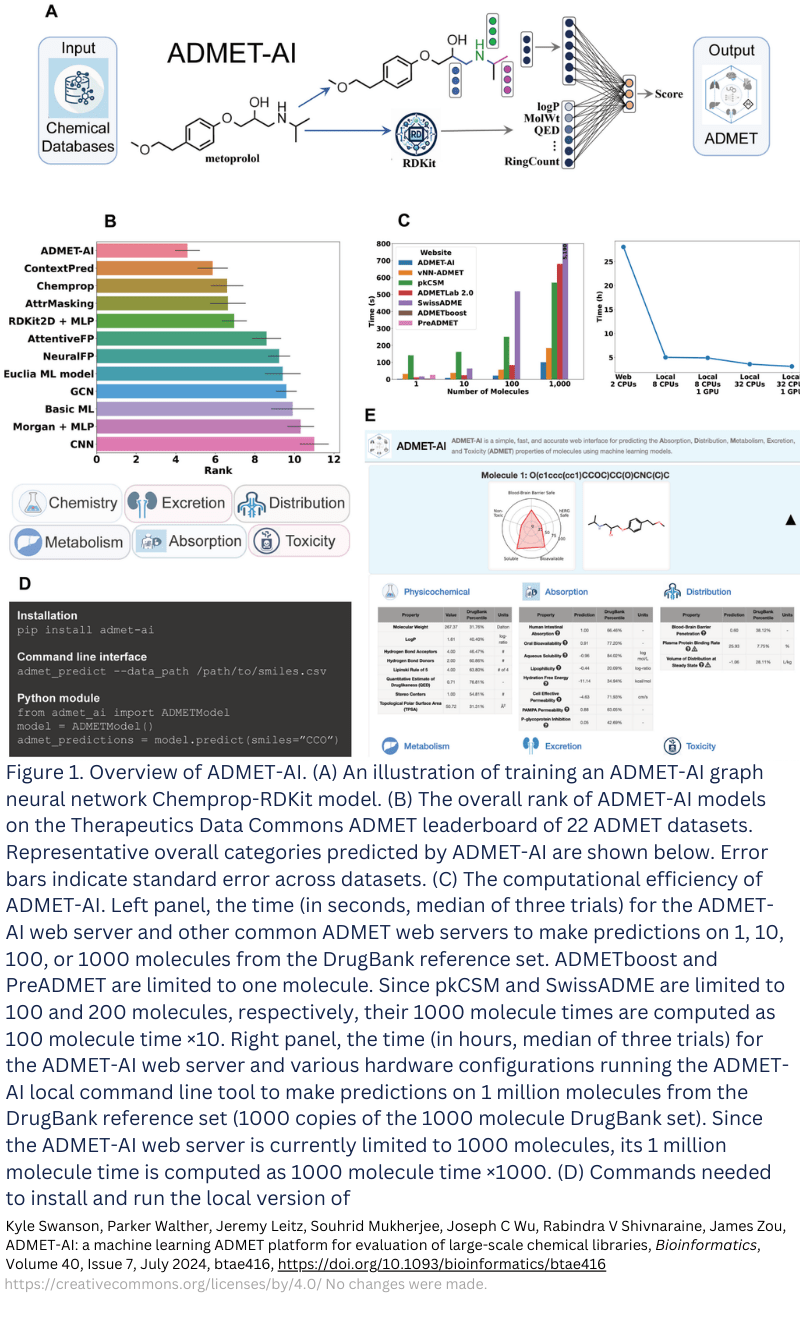
ADMET affects safety and is an important factor in animal research and Phase 1 trials. A Phase 1 study, which is often conducted in healthy volunteers, aims to establish safety before efficacy is evaluated in patients in Phase II. Having a good ADMET prediction can improve the selection of preclinical candidates (PCCs) before Phase 1 is initiated. This prediction can be done with AI, like with ADMET-AI [2]. The selection of PCC can be further improved with Phase 0, a rapid clinical study in the patient population, in which multiple candidates can be administered to compare their properties. At TRACER, we’re experienced in such studies, so the lead candidate can be selected based on in-patient data rather than animal data.
DDI and DTI with Deepdrug
Drug-drug interactions (DDIs) and drug-target interactions (DTIs) are also important aspects of clinical development. AI tool Deepdrug can be used for predicting both negative and positive DDIs and DTIs. As it is more focused on already approved drugs, it can also be used for drug repurposing, repositioning, and providing information regarding polypharmacy. The latter not only informs clinical trials, but it also prevents market withdrawal from adverse drug reactions from negative DDIs [3]. Taken that the market approval rate for drug repurposing is around 30% [4], accelerating the search for drug-target relations with AI can truly benefit patients. Especially rare diseases that lack commercial viability for traditional drug development.
How does AI develop drugs?
How does AI develop drugs? AI is used to discover and validate new targets and improve disease understanding. The found targets, mechanisms, and compounds serve as input for virtual screening and prediction of binding, pharmacodynamics, and ADMET properties. It doesn’t end there; some AI’s, like Iktos, can even design molecular structures and process them in a robotic lab. The paper, The future of pharmaceuticals: artificial intelligence in drug discovery and development [5], provides a technical view of how it works.
Insilico drug discovery candidate selection
Insilico drug discovery is enabling researchers to generate and validate candidates against targets. The impact of variations and mutations in the genome on the disease, the 3D structures of proteins, the binding of novel proteins to targets, and multi-omics data is combined for an accurate prediction. [6, 7, 8, 9, 10].
AI is accelerating drug discovery at an unprecedented pace
Target identification and small molecule discovery in the traditional sense are extremely time-consuming processes that can take somewhere between 2.5 and 6 years. [11, 1]. How fast is AI powered drug discovery?
- Deep Genomics, an AI therapeutics company, discovered a new target and corresponding drug candidate for Wilson’s disease within 18 months [12];
- InSilico Medicine reported an average timeline of 13 months for PCC nomination, but as the example below shows, some programs are mind-blowingly fast;
- Insilico Medicine, in collaboration with Fosun Pharmaceuticals, had a potential first-in-class small molecule inhibitor targeting QPCTL in less than 40 days [13].
QPCTL: Glutaminyl-peptide cyclotransferase-like protein
Example of Phase 0 for an AI-discovered program
InSilico Medicine and Fosun Pharmaceuticals conducted a Phase 0 study (also known as early Phase 1) to evaluate their AI-discovered drug (ISM001-055, also known as Rentosertib. In this first in-human study, a microdose was administered to eight healthy volunteers to evaluate biodistribution, pharmacokinetics, and safety. A Phase 1 study was also conducted [14], and a Phase 2a trial is simultaneously taking place in China and in the USA [15, 16], with already positive results reported [17].
Study not conducted by TRACER.
TRACER’s Phase 0 seamless integration in AI driven drug discovery
TRACER specializes in early phase clinical trials. Phase 0 allows drug developers to evaluate their compound directly in patients after only a single dose extended toxicity study in rodents. Phase 0 can be conducted prior to large animal studies and Phase 1 healthy volunteer studies by using a microdose. A microdose is sufficient for sensitive analytical methods, like PET. By radiolabeling the study drug, you can obtain early pharmacokinetic (PK) and biodistribution (BD) data. When labeled under good manufacturing practice (GMP) conditions, you don’t need a GMP drug substance, lowering the time and cost. After Phase 0, a go/no-go decision can be made on patient data rather than animal data. The obtained patient data can be used to validate findings in subsequent animal and healthy volunteer studies.
A new approach: artificial intelligence drug discovery and development
Artificial Intelligence for drug development is introducing a completely new approach. It is already seen as a successor to the target-based approach (used for approximately the past 30-40 years) that has come under increasing criticism due to its high failure rate. But can AI change the high failure rate in drug development?
Can AI change the high failure rate in drug development?
The failure rate of drug development (from Phase 1 clinical trials to approval) has been around 90% for years [18]. However, this needs to be nuanced. Depending on the criteria, the overall success rate is somewhere between 7.9-14,3% [19]. For some indications like oncology, it’s lower, in the 3,5-5% region [20]. What’s the effect of AI? To date, no AI drugs received approval, so drug development success rates can only be compared in early phases of clinical trials. From Phase 1-2, a success rate of 80%–90% has been reported for AI-developed drugs [1]). This is already a big increase compared to around 52%-64% without AI [19]. Note: the data is very limited. The success rate is based on 24 AI-discovered drugs in Phase 1 trials.
AI drug development example
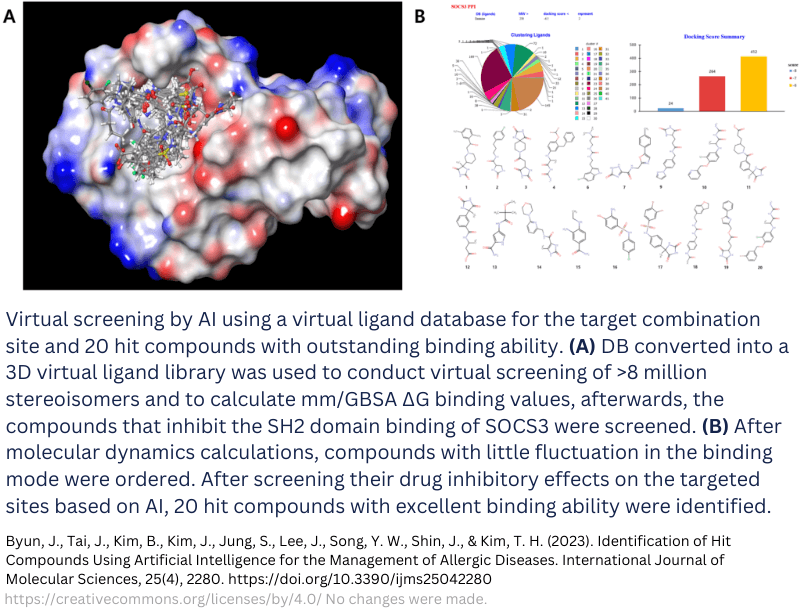
A good example is the development of several hit compounds for allergic disease management. Researchers employed an AI-assisted library for 3D virtual ligands of the target protein. An initial screening of over 8 million stereoisomers was performed. This narrowed down the selection to around 200 compounds. Optimizing this selection for binding affinity to the domain of interest on the target resulted in 20 hit compounds. This approach allowed them to perform high-throughput virtual screening of the binding site in high volumes, quickly resulting in a small selection of compounds that showed selective inhibition in vitro and haltered local allergic responses in in vivo mouse models [21]. The dataset from Enamine was used and processed with software from Schrödinger.
AI drug discovery companies collaborating with pharma
AI drug discovery companies have shown to be effective in discovering new targets and compounds. For further development, they often collaborate with pharmaceutical companies. In the list of pharmaceutical companies using AI for drug discovery, you find many collaborations. To name a few:
- Sanofi (Exscientia AI, Atomwise (Numerion Labs), Owkin, Insilico Medicine, Healx, Earendil Labs, and their own “plai” developed with Aily Labs);
- Astrazeneca (BenevolentAI, Algen Biotechnologies, their own Milton AI, and a collaborating with the Chinese CSPC Pharmaceuticals Group Limited;
- Pfizer (XtalPi);
- Eli Lilly (XtalPi, In Silico, Schrödinger, and their own “AI Factory”).
Top AI drug discovery companies
As you already see, there are many AI drug development companies. Below, we list some of the top AI drug discovery companies and platforms.
Google DeepMind and similar platforms
- AlphaFold is an AI capable of predicting protein structure and folding. Together with the European Molecular Biology Laboratory- European Bioinformatics Institute (EMBL-EBI), it’s also available as AlphaFold DB, a protein structure database.
- DeepMind AlphaProteo designs new proteins aiming to bind to a specific target.
- DeepMind AlphaGenome is used to increase the understanding of the extensive genome, both coding and non-coding regions.
- DeepMind AlphaMissense is based on AlphaFold2 and can be used to predict if changes in DNA sequence and the effects on proteins are estimated to be harmless or pathogenic.
- DeepMind Isomorphic labs is also one of the AI in drug discovery companies from Alphabet (Google). It applies principles of information science to biology with an AI-first approach for drug discovery.
- OpenFold, a re-implementation of DeepMind’s AlphaFold by OpenFold Consortium and the Alquraishi Laboratory.
- SeedFold, ByteDance’s folding model, which, as they state themselves, outperforms AlphaFold3.
Milton AI can reuse data and recognize patterns
Machine learning on big data can lead to an improved disease understanding. An example of this is AstraZeneca’s platform Milton (MachIne Learning with phenoType associatiONs). Milton is an AI able to accurately predict undiagnosed diseases from routine patient data [22]. This is opening the door to investigating treatment of a disease at an earlier stage, based on newly discovered biomarkers. Milton can be accessed publicly.
BenevolentAI
BenevolentAI is a great example of AI in pharma R&D, with drug candidates discovered via the platform already in clinical trials. For example: Compound BEN8744 showed in a Phase 1 trial (NCT06118385) good tolerability and safety and will be further investigated in a Phase 2 trial for ulcerative colitis [23]. Via collaborations, such as BenevolentAI Astrazeneca, new discoveries are validated and developed further [24]. Other compounds from BenevolentAI in clinical trials include BEN-2001 (NCT03194217), BEN-2293 (NCT04737304), and, an IND ready compound, BEN-34712.
Bytedance drug discovery
The most remarkable example of tech companies involved in pharma is ByteDance. The parent company behind social media platform TikTok, making its move into drug discovery with its company Anew Therapeutics [25]. Unknown is how their pharmaceutical endeavor will intertwine with their social media platform. It’s speculated that data from the platform, users discussing medical conditions and treatment, may be used to develop and eventually market new drugs [26]. Anew Therapeutics is not the only step into this field. While a clear list of ventures on ByteDance’s website is missing, other projects that can be linked to ByteDance include CryoSTAR, Amcare hospitals, and health app Xiaohe.
Insilico Medicine AI
When discussing the role of AI in drug discovery and development, Insilico AI is worth mentioning. You may know this company from PandaOmics (Insilico Medicine’s Pharma.ai). The name already indicates it’s not just about protein folding, but includes all kinds of data. Its use is described in papers, discussing how it can be used [27], like for age-related biology [28], targets and drug discovery [15], and an AI-run robotics lab. The company has many drugs already in clinical development and many programs available for licensing. Partnerships with pharmaceutical companies include Eli Lilly, Pfizer, Qilu Pharmaceutical, Hygtia Therapeutics, Servier, Hisun Pharmaceutical, TaiGen Biotechnology, Atossa Therapeutics, Mabwell Bioscience, Huadong Medicine, Harbour BioMed, Therasid Bioscience, and Tenacia biotechnology.
Recursion and Exscientia AI
Is Exscientia now Recursion? Yes, Exscientia AI was acquired by Recursion in late 2024. The joined company has more than 10 advanced programs, some already in Phase 1/2 trials.
Partners include major names like Sanofi, Bayer, Roche, Takeda, Merck, and BMS. The Sanofi Exscientia drug discovery collaboration alone is scheduled to include 15 programs, of which the fourth was reported in May 2025 to have already moved into the lead optimization phase [29].
Iktos
Iktos enables an AI-driven DMTA (Design-Make-Test-Analysis), where the compound moves from discovery and plausible synthetic routes for manufacturing with AI into creating and testing the materials using robotics. With this, preclinical candidates can be delivered in less than two years. In collaborations with the pharmaceutical industry, we see names earlier mentioned, including Servier, Pfizer, and Bayer, but also others, like Cube biotech, Italfarmaco, Nerviano Medical Sciences, Galapagos, and Zealand Pharma.
Healx AI
Healx AI focuses on rare diseases, from the perspective that AI can be used not only to find the next blockbuster drug, but also to find drugs for diseases affecting a low number of patients. Rare disease drug development is often not financially interesting or feasible in traditional drug development. Healx AI has several compounds in (pre)clinical trials and collaborates with companies like Sanofi, Ono Pharmaceutical, and Vuja De Sciences. Another collaboration worth mentioning is with SCI Ventures, an early stage funding company focused on therapies for spinal cord injury (SCI). The collaboration allows Healx access to their specialized insights and dataset. What this shows is that collaborations with AI companies extend beyond pharmaceutical companies and are beneficial in multiple ways. In this case, funding and access to insights and data.
Schrödinger AI drug discovery
We all know Schrödinger’s cat, but did you know Schrödinger AI? While often named under AI drug discovery companies, it doesn’t see itself as such, identifying more as digital chemistry. Since it was founded in 1990, prior to AI, one can understand. To understand this, their computational platform uses the laws of physics for its in-silico predictions instead of training on big data, which is more common for ML for drug discovery. Their (pre)clinical pipeline is filled with proprietary programs, as well as collaborations with well-known pharmaceutical companies such as Lilly, Novartis, BMS, Takeda, and Gilead. Together with Agios, it has two FDA-approved drugs. According to their website, 20 of the top pharmaceutical companies and more than 750 others use the software.
Algen Biotechnologies
Algen Biotechnologies combines CRISPR gene modulation and AI to discover new drugs. The solution consists of two platforms, AlgenBrain and AlgenCRISPR. The first is using deep learning to understand the drivers of disease at a single-cell gene-expression level. AlgenCRISPR then explores these targets for modulation. AlgenBrain is again used to monitor the effects. The method is precisely described in the literature [30]. Algen has a collaboration with AstraZeneca.
Others
There are many AI drug discovery companies. To expand on this list, and to give you a good starting point for further research, take a look at:
- AbCellera
- Aitia
- BenchSci
- BioSymetrics
- BPGbio
- Centella
- Envisagenics
- EVAXION Biotech A/S
- Illumina
- Insitro
- Model Medicine
- NuMedii
- Numerion Labs, formerly Atomwise drug discovery
- Owkin
- Predictive Oncology
- Relay Therapeutics
- Standigm
- Valo Health
- Verge Genomics
- XtalPI
Regulation for AI in drug discovery
The implementation of AI in drug discovery is not without risk. The training data of models is a key component to their success. High-quality, reliable data will need to be fed to deep learning models to make the outcome accurate. The process of medication discovery requires thorough explainability and traceability, not just a black box. In January 2026, the European Medicines Agency (EMA) and Food and Drug Administration (FDA) published their common principles for AI in drug development [31]. And prior, the EMA published its Reflection Paper on the use of AI in the medicinal product lifecycle [32] and the FDA draft guidance Framework to Advance Credibility of AI Models Used for Drug and Biological Product Submissions [33].
The use of patient data in AI
To harness the power of artificial intelligence in drug discovery and development, access to patient data is necessary [5]. This includes data from medical records, which raises ethical and privacy concerns. For drug development, this data can be anonymized, but to use AI in clinical trials, some reference is needed. AI in clinical trials can be used for patient selection, predictive models based on patient characteristics, remote monitoring, image analysis, synthetic control groups, and the use of so-called digital twins [34, 35]. By improving the selection of participants, monitoring them, and virtually testing treatments on these digital twins of real participants, safety can be improved. With the increasing use of AI in routine care, collecting data might be easier than expected but. Although this is beyond the scope of this article, strict usage must be defined for ethical reasons.
The future of AI in drug discovery and development
What is the future of AI in drug development? The results of new targets and drugs discovered using AI are promising, and the pharmaceutical industry is investing heavily in this area. AI has proven to drastically reduce discovery timelines. More newly AI-discovered compounds are entering clinical trials each year. To prevent overflooding, the industry will need scalable infrastructure that can efficiently de-risk compounds early. Think of organoids, microphysiological systems, organ-on-chip technologies, and Phase 0 clinical trials. AI has revolutionized drug discovery; now it’s time for AI in drug development to keep up. For this, a shift from traditional clinical studies to faster, adaptive, data-driven clinical trials is needed, accompanied by clear regulations and more data.
We at TRACER are happy to discuss how flexible trial designs, Phase 0 and molecular imaging can be applied in your drug development program.
Schedule a meeting
Abbreviations
| ADMET | Absorption, Distribution, Metabolism, Excretion, and Toxicity |
| AI | Artificial intelligence |
| BD | biodistribution |
| DB | Database |
| DDIs | Drug-Drug Interactions |
| DL | Deep Learning |
| DMTA | Design-Make-Test-Analysis |
| DTI | Drug-Target Interactions |
| EMBL-EBI | European Molecular Biology Laboratory- European Bioinformatics Institute |
| EMA | European Medicines Agency |
| FDA | Food and Drug Administration. |
| GET | General Expression Transformer |
| GMP | good manufacturing practice |
| ML | machine learning |
| PCC | preclinical candidate |
| PET | positron emission tomography |
| PK | pharmacokinetic |
| R&D | Research and Development |
| SCI | spinal cord injury |
Citations
DOI: 10.1021/acs.jcim.3c01619 https://pubs.acs.org/doi/10.1021/acs.jcim.3c01619
14 January 2026 https://www.ema.europa.eu/en/news/ema-fda-set-common-principles-ai-medicine-development-0
the medicinal product lifecycle, 9 September 2024 https://www.ema.europa.eu/en/documents/scientific-guideline/reflection-paper-use-artificial-intelligence-ai-medicinal-product-lifecycle_en.pdf
Although this article has been composed with great care and attention, we cannot guarantee its accuracy. If you have any suggestions or additions to this article, please email info@tracercro.com.
No rights can be derived from this publication. This blog does not make claim or promote ownership to any intellectual property, study information, or copyrighted terms wherein.